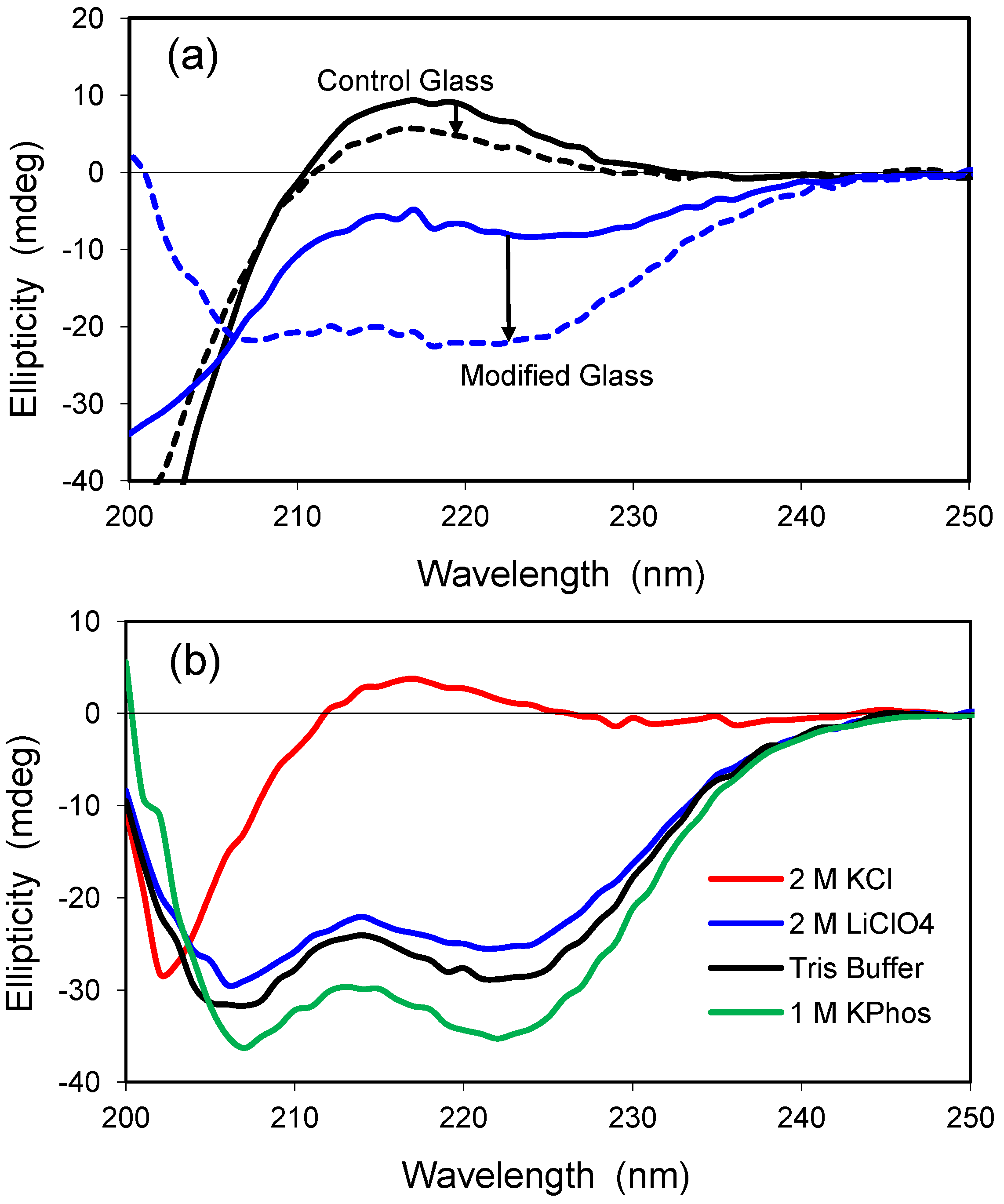
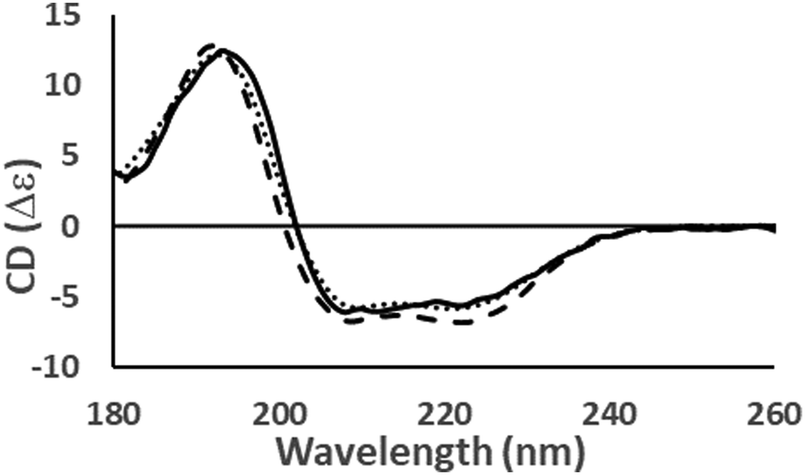
In addition, their spectra before and after treatment with various conditions to produce unfolded or denatured structures were also compared, with the aim of defining the differences between CD spectra of PPII and disordered structures. Their native spectra were compared as representatives of PPII structures. In this study, synchrotron radiation circular dichroism spectroscopy was used to investigate the spectral properties of collagen and polyproline, which both contain PPII‐type structures. However the “other” category can also include polyproline II (PPII)‐type structures, whose spectral properties have not been well‐distinguished from those of unordered structures. Many proteins identified as being “Intrinsically Disordered Proteins” have a significant amount of their structure that is neither sheet, helix, nor turn this type of structure is often classified by CD as “other”, “random coil”, “unordered”, or “disordered”. A.Ĭircular dichroism (CD) spectroscopy is a valuable method for defining canonical secondary structure contents of proteins based on empirically‐defined spectroscopic signatures derived from proteins with known three‐dimensional structures. Distinct circular dichroism spectroscopic signatures of polyproline II and unordered secondary structures: Applications in secondary structure analyses Distinct circular dichroism spectroscopic signatures of polyproline II and unordered secondary.
